[the_ad id=”9653″]
Unlock the secrets to mastering the GPAT exam and catapult your career in pharmacy! Dive into the ultimate resource: the GPAT 2023 Previous Year Question Paper, now accessible on pharmacyindia.co.in. Elevate your preparation with this comprehensive guide, meticulously designed to enhance your understanding of key concepts and exam patterns.
Gain invaluable insights into the structure of the GPAT exam, refine your strategies, and boost your confidence for success. Don’t miss out on this invaluable tool as you embark on your journey towards acing the GPAT exam and unlocking boundless opportunities in the pharmaceutical realm. Visit pharmacyindia.co.in today and pave the way for your triumph!
Also Read: GPAT 2023 Shift-II Previous Year Question Paper
PHARMACEUTICS
| LIST (FORMULATION INGREDIENT) | LIST ( ACTIVITY/ PROPERTY) |
| 1. Salts of d-glucuronic acid polypeptides and amino acids | [P] Are pseudoplastic and plastic in nature |
| 2. Surfactants, both ionic and nonionic | [Q] Form a multimolecular film around the dispersed droplets of oil in an o/w emulsion |
| 3. Magnesium aluminium silicate | [R] Emulsifier belonging to the class of solid particles form W/O emulsion |
| 4. Structured vehicles | [S] Emulsifier belonging to the class of solid particles and form O/W emulsion |
| [T] Have been used to bring about flocculation of suspended particles |
|
INGREDIENT |
FUNCTION |
TABLET A |
TABLET B |
TABLET C |
TABLET D |
||
|
P |
Acetaminophen |
I |
Drug |
300mg |
— |
300mg |
300mg |
|
Q |
Aspirin |
II |
Drug |
— |
300mg |
— |
— |
|
R |
Lactose |
III |
Filler |
100mg |
100mg |
— |
100mg |
|
S |
Avicel |
IV |
Filler |
— |
— |
100mg |
— |
|
|
Starch |
|
Disintegrant |
50mg |
50mg |
— |
50mg |
|
|
Avicel |
|
Disintegrant |
— |
— |
50mg |
— |
|
|
Mag stearate |
|
Lubricant |
2mg |
2mg |
2mg |
2mg |
|
|
Gelatin |
|
Binder |
10mg |
10mg |
10mg |
10mg |
| List I (Schedule) | List II (Cover the) |
| 1. Schedule Y | (P) Shelf life of drugs |
| 2. Schedule FF | (Q) Requirements for clinical trails |
| 3. Schedule O | (R) Disinfectant liquids |
| 4. Schedule P | (S) Ophthalmic ointments |
(a) 2-Pentene
(b) 2-Methyl-2-butene
(c) 1-Pentene
(d) 1-Methyl-2-butene
44. The following are the processes occurring during flame atomization in atomic absorption spectrometry
[P] Volatilization [Q] Ionization [R] Nebulization
[S] Desolvation [T] Dissociation
Arrange the processes in sequential order and choose the correct answer from below
(a) R, Q, P, S, T
(b) R, S, T, Q, P
(c) P, R, S, Q, T
(d) R, S, P, T,Q
45. A mixture of p-anisaldehyde and formaldehyde in the presence of concentrated sodium hydroxide results in
(a) p-Methoxy sodium benzoate
(b) Sodium-p-methoxy benzene
(c) P-Methoxy benzyl alcohol
(d) p-Methoxy benzoyl alcohol
46. Which one of the following compound is a precursor for the biosynthesis of cholesterol
(a) Progesterone
(b) Lanosterol
(c) Cholic acid
(d) Coprostanol
47. Which one among the following drugs has the IUPAC name, α, α, α, α- tetramethyl 5- (1 H-1,2,4-triazole- 1-ylmethyl)-1,3-benzenediacetonitrile
(a) Letrozole
(b) Anastrozole
(c) Exemestane
(d) Aminoglutethimide
48. The reaction between naphthalene and chromium trioxide in the presence of glacial acetic yields
(a) Naphthalene-1,4-dione
(b) 4-Hydroxynaphthalen-1(4H)-one
(c) Naphthalene-1.2-dione
(d) 1-Hydroxynaphthalen-2(1H)-one
49. Platinum electrode surrounded by an outer tube, in which hydrogen passes entering through side inlet and escaping at the bottom through the test solution is called as
(a) Silver electrode
(b) Calomel electrode
(c) Standard hydrogen electrode
(d) Indicator electrode
50. Which of the following gives correct rank order from fastest to slowest of the relative rates in SN reaction of methyl bromide, tert-butyl bromide, isopropyl bromide and ethyl bromide
(a) Methyl bromide > Ethyl Bromide > Isopropyl bromide > tert-Butyl bromide
(b) tert-Butyl bromide > Isopropyl bromide > Ethyl Bromide > Methyl bromide
(c) Ethyl bromide > Methyl Bromide > Isopropyl bromide > tert-Butyl bromide
(d) Methyl bromide > Ethyl Bromide > tert-Butyl bromide > Isopropyl bromide
51. Select the correct set of anticancer drugs that belong to “Pyrimidine and related compounds”
(a) 5-Fluorouracil, Tegafur, Decitabine, 5-Azacytidine
(b) 5-Fluorouracil, Tegafur, Decitabine, Clofarabine
(c) Tegafur, Decitabine, 5-Azacytidine, Clofarabine
(d) Tegafur, Decitabine, 5-Azacytidine, Pentostatin
52. E2 elimination converts neomenthyl chloride into a mixture of the following compounds
(a) 3-menthene (75%) and 2-menthene (25%)
(b) 4-menthene (75%) and 2-menthene (25%)
(c) 3-menthene (25%) and 5-menthene (75%)
(d) 1-menthene (50%) and 3-menthene (50%)
53. Ethyl-2 (p-chlorophenoxy)-2-methyl propionate is IUPAC name of
(a) Fenofibrate
(b) Colestipol
(c) Clofibrate
(d) Colesevelam
54. Identify the vibrational modes shown by sulfur dioxide molecule in IR spectroscopy
(a) Symmetric stretching and asymmetric stretching
(b) Symmetric stretching and scissoring
(c) Asymmetric stretching and scissoring
(d) Symmetric stretching, asymmetric stretching and scissoring
55.
 Select the correct product A of the above-given reaction from the four choices given below
Select the correct product A of the above-given reaction from the four choices given below 56. The perfect orientation for a Diels-Alder reaction between the reactants is
56. The perfect orientation for a Diels-Alder reaction between the reactants is(a) Diene should be S-cis and reaction endo facing
(b) Diene should be S-cis and reaction exo facing
(c) Diene should be S-trans and reaction endo facing
(d) Diene should be S-trans and reaction exo facing
57. How would you prepare 2000 mL of 0.15 M NaOH aqueous solution
(a) Dissolve 12 g of NaOH in distilled water and dilute to 2000 mL
(b) Dissolve 15 g of NaOH in distilled water and dilute to 2000 mL
(c) Dissolve 10 g of NaOH in distilled water and dilute to 2000 mL
(d) Dissolve 7.5 g of NaOH in distilled water and dilute to 2000 mL
58. Identify what is not a continuum source of radiation for use in absorption and fluorescence spectrophotometers
(a) Argon are lamp
(b) Hollow-cathode lamp
(c) Deuterium lamp
(d) Xenon are lamp
59. The molecular formula of purine is
(a) C5H4N4
(b) C6H5N3
(c) C7H6N2
(d) C4H3N5
60. Given below are two statements, one is labelled as Assertion and the other is labelled as Reason
Assertion (A) The disadvantage of atomic absorption spectroscopy is the need for each element to be analysed
Reason (R) As atomic absorption spectrophotometer uses different halo cathode lamp for each element, it is very specific for an individual element under test
In light of the above statements, choose the correct answer from the options given below
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is NOT the correct explanation of A
(c) A is true but R is false
(d) A is false but R is true
61. In context to voltammetry, which of the following statement is false
(a) Technique can be used to analyse organic compounds containing carbonyl groups
(b) Organic solvents cannot be used as aqueous organic mixture in this technique
(c) Immuno sensors are available in voltammetry
(d) Triangular waveform is used for excitation of solution in cyclic voltammetry
62. Choose the correct order of decreasing dielectric constant
(a) Water > Formamide > Methanol > Acetone
(b) Water > Methanol > Acetone > Formamide
(c) Formamide > Acetone > Methanol > Water
(d) Formamide > Water > Methanol > Acetone
63. How many optical isomers are possible for lactic acid
(a) 2 (b) 4 (c) 6 (d) 8
64. Predict the wavelength of absorption band in the UV spectrum of the above shown structure
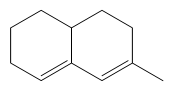 (a) 252 nm (b) 248 nm (c) 244 nm (d) 240 nm
(a) 252 nm (b) 248 nm (c) 244 nm (d) 240 nm65. Electrophilic substitution reaction of pyridine, when carried out in the presence of KNO3 and conc. H2SO4 at 300°C, leads to the formation of
(a) 4-Nitropyridine
(b) 3-Nitropyridine
(c) 2-Nitropyridine
(d) N-nitro pyridinium salt
66. A reagent may attach itself to a conjugated diene to the carbons at the two ends of the conjugated system. Identify the reaction involved from the following
(a) 1,2-addition
(b) 1,4-addition
(c) 1,3-addition
(d) 2,4-addition
67. Following groups exert anyone of the effects on electrophilic aromatic substitution. Identify whether all three are
(a) Weakly activating
(b) Deactivating
(c) Moderately activating
(d) Strongly activating
68. The correct rank order of orientation of sulfonation in toluene is
(a) 4-methylbenzenesulfonic acid > 2-methylbenzenesulfonic acid > 3 methylbenzenesulfonic acid
(b) 2-methylbenzenesulfonic acid > 3-methylbenzenesulfonic acid > 4-methylbenzenesulfonic acid
(c) 3-methylbenzenesulfonic acid > 4-methylbenzenesulfonic acid > 2-methylbenzenesulfonic acid
(d) 3-methylbenzenesulfonic acid > 2-methylbenzenesulfonic acid > 4-methylbenzenesulfonic acid
69. Lucas test is very rapid with
(a) 1°alcohol
(b) 2°alcohol
(c) 3°alcohol
(d) Phenol
70. As the solution of a strong electrolyte is diluted, the following phenomenon is observed
(a) The specific conductance decreases and equivalent conductance increases
(b) The specific conductance increases and equivalent conductance decreases
(c) Both specific conductance and equivalent conductance increase
(d) Both specific conductance and equivalent conductance decrease
71. The molecule having zero dipole moment is
(a) BF3 (b) HF (c) NH3 (d) CH3C1
(a) Haloperidol
(b) Clozapine
(c) Thioridazine
(d) Fluphenazine
73. Following is not an example of Carbapenem
(a) Thienamycin
(b) Imipenem
(c) Piperacillin
(d) Meropenem
74. Which of the following Urinary Tract Anti-Infective agents requires an acidic pH of urine for optimum action
(a) Gentamicin
(b) Erythromycin
(c) Carbenicillin
(d) Streptomycin
75. Which of the following drugs has an apparent volume of distribution approximately 6500 litres
(a) Amoxicillin
(b) Ibuprofen
(c) Chloroquine
(d) Diazepam
76. Match the following concept in List I with parameters in List II
| List I (Concept) | List II (Parameter) |
| 1. Volume of Distribution | (P) Measure volume of real physiological plasma |
| 2. Evans Blue | (Q) Human serum albumin |
| 3. 3.5-5% | (R) Volume of blood |
| 4. Metallothionein | (S) Ratio of body drug content to plasma concentration |
| (T) Protein present in kidney to bind metal |
Choose the correct answer from the options given below
(a) 1-[P], 2-[Q], 3-[R], 4-[S]
(b) 1-[T], 2-[S], 3-[R], 4-[Q]
(c) 1-[S], 2-[P], 3-[Q], 4-[T]
(d) 1-[R], 2-[Q], 3-[S], 4-[T]
77. Trastuzumab is a/an
(a) EGFR/HER2 inhibitor
(b) Angiogenesis inhibitor
(c) EGF receptor (HER1) inhibitor
(d) BCR-ABL tyrosine kinase inhibitor
78. When is a New Drug Application (NDA) made
(a) Once the animal studies are done and drug is declared safe in animals
(b) Once the animal studies are done and drug is declared safe and effective in animal studies
(c) After the phase III clinical trials
(d) After the phase IV clinical trials
79. Identify the drug that is not among the drugs recommended as first time drug in the treatment of Partial seizers with or without generalization
(a) Carbamazepine
(b) Valproate
(c) Diazepam
(d) Lamotrigine
80. Which of the following hyperlipidemic drugs act via a GPCR
(a) Nicotinic acid
(b) Fenofibrate
(c) Atorvastatin
(d) Ezetimibe
81. All the following are TNF-α Inhibitors EXCEPT
(a) Etanercept
(b) Infliximab
(c) Adalimumab
(d) Basiliximab
82. Match the following cells of immune system List I with their functions List II
| List I (Immune System) | List II (Functions) |
| 1. Mast cells | (P) Master of Immune Systems |
| 2. Lymphocytes | (Q) Allergic Reactions |
| 3. T-cells | (R) Cell mediated immune reactions |
| 4. Monocytes-macrophages | (S) Antigen recognition, Phagocytosis |
Choose the correct answer from the options given below
(a) 1-[P], 2-[Q], 3-[R], 4-[S]
(c) 1-[R], 2-[P], 3-[Q], 4-[S]
83. Which of the following drug can produce mydriasis without Cycloplegia
(a) Atropine
(b) Tropicamide
(c) Homatropine
(d) Ephedrine
84. What do you mean by Orphan drug
(a) A drug meant to be distributed among the orphans who can not afford the cost of the drug
(b) A drug for a disease which is not having any other treatment options at all
(c) A drug which is useful for rare disease
(d) A drug that is available in abundance
85. Normal value of HbA1c falls in the range of
(a) 0.1% to 0.8%
(b) 6.5% to 7.5%
(c) 21.5% to 24.5%
(d) 51.5% to 53.5%
86. A 70 kg woman was administered 1000 mg of the drug as i.v bolus. After its uniform distribution in the body, the plasma concentration of the drug was found to be 50 mg/
L. What is its volume of distribution
(a) 70L (b) 500L (c) 20L (d) 0.1L
87. A hypertensive patient receiving a drug ‘Y’ for managing BP was prescribed a tricyclic antidepressant. As a result, there was an abolition of the antihypertensive action of ‘Y’. Which of the following drug could be ‘Y’
(a) Atenolol
(b) Captopril
(c) Clonidine
(d) Diltiazem
88. Put the events of acute inflammation in proper sequence
(a) Accumulation of fluid and plasma at the affected site → intravascular activation of plate lets → polymorphonuclear neutrophils → followed by healing
(b) Polymorphonuclear neutrophils → accumulation of fluid and plasma at the affected site
→ intravascular activation of platelets → followed by healing
(c) Accumulation of fluid and plasma at the affected site → polymorphonuclear neutrophils
→ intravascular activation of platelets → followed by healing
(d) Intravascular activation of platelets → polymorphonuclear neutrophils → accumulation of fluid and plasma at the affected site → followed by healing
89. Which of the following is NOT an example of an mTOR inhibitor
(a) Everolimus
(b) Tacrolimus
(c) Temsirolimus
(d) Sirolimus
90. Alzheimer’s disease is mainly because of the neurodegeneration of the following part of the brain
(a) Hypothalamus
(b) Midbrain
(c) Hippocampus
(d) Cerebellum
91. Which of the following has been found to act as a male contraceptive without affecting libido and potency
(a) Cyproterone
(b) Gossypol
(c) Centchroman
(d) Goserelin
92. Choose the most appropriate statement for the Peptic ulcer disease caused by NSAIDs
(a) H2 antagonists offer rapid healing of ulcer provided the NSAID is discontinued
(b) H2 antagonists offer rapid healing
(c) Proton Pump Inhibitor is to be given only if the NSAID is discontinued
(d) NSAIDs are strictly contraindicated with Proton Pump Inhibitors
93. Lateral geniculate nucleus is associated with
(a) Vision
(b) Hearing
(c) Olfaction
(d) Gustation
94. Which of the following drug used in the chemotherapy of some types of leukemia, satis fies the statements
Statement I: It is effective orally
Statement II: It has near 100% oral bioavailability
In light of the above statements, choose the correct answer from the options given below
(a) Asparaginase
(b) Doxorubicin
(c) Mitomycin
(d) Hydroxyurea
95. Statement I: In vasospastic angina, the imbalance occurs when the myocardial oxygen requirement increases, as during exercise, and coronary blood flow does not increase proportionately.
Statement II: In Prinzmetal’s angina, oxygen delivery decreases as a result of reversible coronary vasospasm
In light of the above statements, choose the most appropriate answer from the options given below
(a) Both Statement I and Statement II are correct
(b) Both Statement I and Statement II are incorrect
(c) Statement I is correct but Statement II is incorrect
(d) Statement I is incorrect but Statement II is correct
96. In ECG, the P wave corresponds to the following event
(a) Atrial depolarisation
(b) Ventricular depolarisation
(c) Atrial repolarisation
(d) Ventricular repolarization
97. Which of the following cranial nerve helps in accommodating the eye for near vision
(a) Oculomotor
(b) Optic
(c) Trochlear
(d) Facial
98. Which of the following is true for bone tissue cell differentiation and maturation
(a) Osteogenic → Osteoblasts → Osteocytes
(b) Osteogenic → Osteoblasts → Osteoclasts
(c) Osteocytes → Osteogenic → Osteoblasts
(d) Osteoclasts → Osteoblasts → Osteocytes
99. A drug ‘X’ is more selective for the α1 subunit of BZD receptors. It lacks effect on slow- wave sleep. minimum residual daytime sedation, no rebound insomnia on discontinuation, low abuse potential. Which of the following can be ‘X’
(a) Flurazepam
(b) Flumazenil
(c) Melatonin
(d) Zolpidem
100. Which of the following is a fourth-generation cephalosporin
(a) Ceftriaxone
(b) Cefaclor
(c) Cefuroxime
(d) Cefepime
101. Which of the following metabolite is used to inactivate the vasicotoxic metabolites leading to hemorrhagic cystitis by alkylating agents used in the treatment of cancer
(a) Acroline
(b) Aldophosphamide
(c) Cyclophosphamide
(d) Mesna
102. All the following are true with Metoclopramide EXCEPT
(a) 5-HT1 receptor antagonist
(b) D receptor antagonist
(c) 5-HT3 receptor antagonist
(d) 5-HT4 receptor agonist
103. Given below are two statements
Statement I: Levodopa is metabolized peripherally but capable of crossing Blood Brain Barrier, thus a best drug in treating Parkinsonism
Statement II: Carbidopa crosses Blood Brain Barrier, thus a best combination for pro tecting levodopa in CNS
In light of the above statements, choose the correct answer from the options given below
(a) Both Statement I and Statement II are true
(b) Both Statement I and Statement II are false
(c) Statement I is true but Statement II is false
(d) Statement I is false but Statement II is true
104. Which of the following is a tetracycline antidepressant that has additional Dopamine D2 receptor blocking and neuroleptic properties as well as a greater tendency to cause seizure in overdose [DROPPED QUESTION]
(a) Dothiepin
(b) Doxepin
(c) Trazodone
(d) Amoxapine
105. Which one is the right sequence of the intermediates in the biosynthesis of opium alkaloids
I. Tyrosine II. Reticuline III. Codeine
IV. Morphine V. Thebaine
Click Here to join WhatsApp Group
Choose the correct answer from the options given below
(a) I, II, III, IV, V
(b) I, II, V, III, IV
(c) I, II, V, IV, III
(d) I, II, IV, V, III
106. Arrange the following intermediates in the synthesis of isoprenoids in the right sequence
I. Squalene II. Farnesyl PP III. Geranyl PP
IV. Acetyl CoA V. Mevalonate
Choose the correct answer from the options given below
(a) IV, II, III, I, V
(b) IV, V, III, II, I
(c) II, III, IV, I, V
(d) V, II, III, I, IV
107. If the resins contain benzoic acids or cinnamic acids they are called
(a) Colophony
(b) Balsams
(c) Glycoresins
(d) Resene
108. Lycopodium spores are used in quantitative microscopy for the following
I. Determine % purity of drugs
II. Estimation of percentage of foreign organic matter
III. Determination of palisade ratio
IV. Measurement of area of single layered tissue
Choose the correct answer from the options given below
(a) I, II and III only
(b) I, II and IV only
(c) I only
(d) II and IV only
109. The ring structure present in strychnine alkaloid is
(a) Indole
(b) Purine
(c) Phenanthrene
(d) Imidazole
110. Isabgol belongs to family
(a) Apocynaceae
(b) Plantaginaceae
(c) Solanaceae
(d) Golaceae
Click Here to Join Telegram Group
111. Lignin is a complex polymer which can be stained pink in the tissue using the following chemicals
(a) Chloral Hydrate and Phloroglucinol
(b) Chlor-Zinc-Iodine
(c) Phloroglucinol and hydrochloric acid
(d) Chloral Hydrate, Zinc and Ammonia
112. Match the following Ayurvedic formulations under Column I with the process/ properties under Column II and choose the correct options
| List I (Ayurvedic Formulations) | List II (Process or Property) |
| 1. Bhasma | (P) Semisolid |
| 2. Arista | (Q) Calcination |
| 3. Churna | (R) Alcohol generation |
| 4. Lehya | (S) Dry Powder |
| (T) Decoction |
Choose the correct answer from the options given below
(a) 1-[P], 2-[R], 3-[S], 4-[T]
(b) 1-[R], 2-[Q], 3-[P], 4-[S]
(c) 1-[R], 2-[T], 3-[P], 4-[Q]
(d) 1-[Q], 2-[R], 3-[S], 4-[P]
113. Match the types of glycosides under Column I with their respective example under Column II and choose the correct option
| Column I (Glycosides) | Column II (Examples) |
| 1. Anthracene | (P) Digitalis |
| 2. Cardiac | (Q) Liquorice |
| 3. Saponin | (R) Senna |
| 4. Cyanogenetic | (S) Ashwagandha |
| (T) Bitter Almond |
Choose the correct answer from the options given below
(a) 1-[R], 2-[P], 3-[Q], 4-[T]
(b) 1-[T], 2-[P], 3-[Q], 4-[R]
(c) 1-[S], 2-[Q], 3-[P], 4-[R]
(d) 1-[Q], 2-[R], 3-[S], 4-[P]
114. Quinine and Quinidine differs in
(a) Chemical nature
(b) Molecular formula
(c) Rotating the plane of polarized light
(d) Precursor of biosynthesis
Subscribe our PHARMACY INDIA Youtube Channel for more Pharma Updates
(a) Polysaccharide
(b) Protein
(c) Nucleic acid
(d) Peptide
116. Which one is not the characteristics of the Hexose Monophosphate Pathway
(a) It produces CO2
(b) It requires ATP for phosphorylation
(c) It is controlled by inhibition of glucose-6 phosphate dehydrogenase by NADPH
(d) It produces ribose-5-phosphate
117. Match List I with List II
| Column I (Name of Vitamin) | Column II (Functions of Vitamins) |
| 1. Riboflavin | (P) The electron acceptor for isocitrate dehydrogenase |
| 2. Niacin | (Q) Decarboxylation of α-ketoglutarate |
| 3. Thiamine | (R) Part of Co-enzyme A |
| 4. Pantothenic acid | (S) Cofactor for succinate dehydrogenase |
| (T) Enzyme activity regulator such as protein kinase C |
(a) 1-[S], 2-[P], 3-[Q], 4-[R]
(b) 1-[R], 2-[Q], 3-[S], 4-[T]
(c) 1-[P], 2-[R], 3-[S], 4-[Q]
(d) 1-[Q], 2-[T], 3-[P], 4-[S]
118. DNA and RNA contain the following two major purine bases
(a) Guanine and Cytosine
(b) Adenine and Guanine
(c) Thymine and Uracil
(d) Adenine and Uracil
119. Match the process of reproduction and genetic exchange under column I with the explanation under column II Match List I with List II
| List I (PROCESS OF REPRODUCTION AND GENETIC EXCHANGE) | List II (EXPLANATION) |
| 1. Binary fission | (P) Transfer of genetic material from the donor to recipient bacterium through cell contact |
| 2. Transformation | (Q) Common vegetative reproduction |
| 3. Transduction | (R) Transfer of genetic material in bacteria through virus |
| 4. Conjugation | (S) Horizontal gene transfer by taking up of foreign genetic material (naked DNA) |
Choose the correct answer from the options given below
(a) 1-[P], 2-[R], 3-[S], 4-[Q]
(b) 1-[R], 2-[P], 3-[S], 4-[Q]
(c) 1-[Q], 2-[S], 3-[P], 4-[R]
(d) 1-[Q], 2-[S], 3-[R], 4-[P]
120. Match List I with List II
| Column I (FERMENTATION PRODUCTS) | Column II (STRAIN USE) |
| 1. Dextran | (P) Clostridium tetani |
| 2. Bacterial amylase | (Q) Brevibacterium sp. |
| 3. Glutamic acid | (R) Leuconostoc mesenteroides |
| 4. Vitamin B12 | (S) Bacillus subtilis |
| (T) Streptomyces olivaceus |
Choose the correct answer from the options given below
(a) 1-[T], 2-[P], 3-[S], 4-[R]
(b) 1-[P], 2-[S], 3-[Q], 4-[R]
(c) 1-[Q], 2-[R], 3-[T], 4-[S]
(d) 1-[R], 2-[S], 3-[Q], 4-[T]
121. Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A : Exotoxins diffuse freely through the bacterial cell wall into the medium in which the organisms are growing
Reason R : They are water soluble and can pass into the surrounding medium
In the light of the above statements, choose the correct answer from the options given below
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is NOT the correct explanation of A
(c) A is true but R is false
(d) A is false but R is true
122. Basic dyes are used in microbiological staining. Which of the following statement is wrong about mechanism of staining
(a) The positive ions on the surface of bacteria form a colored complex with the dye
(b) Ionic exchange between the negative charge on the bacteria and positive charge of dye takes place
(c) Ionic exchange between the positive charge on the bacteria and negative charge of dye takes place
(d) The neutral charge on the surface of bacteria forms a colored complex with the basic dye
123. Which of the following represents a complete list of products that are offered by a com pany for Sale
(a) Product Variety
(b) Product Mix
(c) Product Item
(d) Product Line
124. Shift from ‘Try my product’ to ‘Prefer my brand’ marketing strategy is done at what stage of product life cycle
(a) Introduction
(b) Growth
(c) Maturity
(d) Decline
125. Haemophilus influenzae type B (Hib) vaccine is an example of
(a) Subunit vaccine
(b) Toxoid vaccine
(c) Recombinant protein vaccine
(d) Conjugate vaccine
|
1-b |
2-a |
3-d |
4-b |
5-c |
6-d |
7-c |
8-a |
9-a |
10-a |
|
11-a |
12-d |
13-c |
14-a |
15-d |
16-c |
17-c |
18-b |
19-c |
20-d |
|
21-a |
22-b |
23-b |
24-d |
25-a |
26-d |
27-a |
28-a |
29-a |
30-b |
|
31-d |
32-b |
33-d |
34-c |
35-b |
36-c |
37-c |
38-b |
39-b |
40-d |
|
41-c |
42-d |
43-b |
44-d |
45-c |
46-b |
47-b |
48-a |
49-c |
50-a |
|
51-a |
52-a |
53-c |
54-a |
55-d |
56-a |
57-a |
58-b |
59-a |
60-c |
|
61-b |
62-d |
63-a |
64-d |
65-b |
66-b |
67-c |
68-a |
69-c |
70-a |
|
71-a |
72-b |
73-c |
74-c |
75-c |
76-c |
77-a |
78-c |
79-c |
80-a |
|
81-d |
82-b |
83-d |
84-c |
85-b |
86-c |
87-c |
88-a |
89-b |
90-c |
|
91-b |
92-b |
93-a |
94-d |
95-d |
96-a |
97-a |
98-a |
99-d |
100-d |
|
101-d |
102-a |
103-c |
104-* |
105-b |
106-b |
107-b |
108-c |
109-a |
110-b |
|
111-c |
112-d |
113-a |
114-c |
115-a |
116-b |
117-a |
118-b |
119-d |
120-d |
|
121-a |
122-d |
123-b |
124-b |
125-d |
|
||||
<<<<<<<<<<<<JOIN US>>>>>>>>>>>>>>>>
| Subscribe our PHARMACY INDIA Youtube Channel for more Pharma Updates | Click Here |
| Follow us on Instagram | Click Here |
| Download PHARMACY INDIA MOBILE APP from Google Play Store | Click Here |
| Follow us on LinkedIn | Click Here |




